
Research
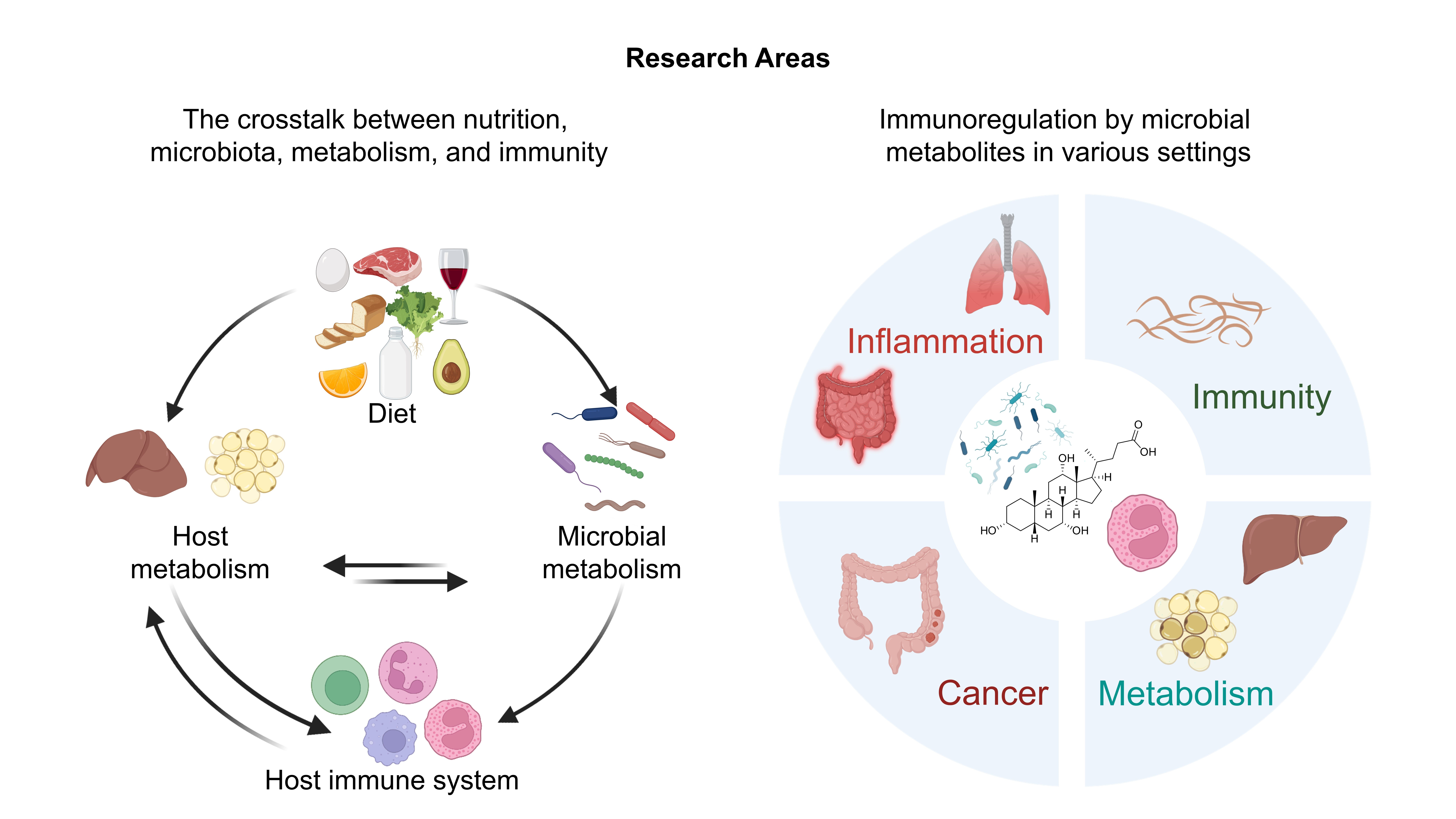
Diet plays a pivotal role in shaping not only our own metabolism but also that of our gut microbiota. The nutritional status of the host can dramatically alter both the composition and metabolic activity of intestinal microbiota, which, in turn, profoundly effects host immune and metabolic outcomes. The gut microbiota produce a vast array of bioactive small-molecule metabolites that regulate diverse physiological processes in the host. However, the majority of these microbial metabolites, their regulation by diet, and their effects on host immunity and metabolism remain largely unexplored.
To address these gaps, we have adopted a cutting-edge metabolomics-based approach that integrates targeted and untargeted metabolomics with bacterial genetics and host transcriptomics. This comprehensive strategy enables us to pinpoint key microbial metabolites, identify the microbial genes responsible for their production, and uncover the host receptors that mediate their effects on host physiology. Through this lens, we seek to unravel how specific nutrients influence the gut microbiota and how these diet-microbiota interactions shape host immune and metabolic responses. Moreover, we are investigating the role of interconnected systems, such as the nervous system, in these regulatory networks.
Our lab employs flow cytometry and microscopy to explore the interactions of various immune cells, such as eosinophils, with neighboring non-immune cells like epithelial cells, stromal cells, and neurons. Additionally, we leverage metagenomic analyses, single-cell and spatial transcriptomics, and high-resolution mass spectrometry-based metabolomics to decode the immunoregulatory functions of microbiota-derived metabolites and their host receptors. The ultimate aim of our research is to uncover the molecular mechanisms through which environmental factors, including diet, shape immune responses. These insights will pave the way for designing novel interventions and therapeutic strategies to prevent and treat disease.
The ongoing efforts in the lab are focused on defining the impact of diet and microbial metabolites on:
- intestinal inflammation and tumorigenesis.
- immune-metabolic interactions in obesity and liver disease.
Figure 1.
Bio
Dr. Arifuzzaman earned his Ph.D. in Molecular Genetics and Microbiology from Duke University, where he studied innate immune defense mechanisms of mast cells and macrophages in Dr. Soman Abraham’s lab. He pursued his postdoctoral training with Dr. David Artis at Weill Cornell, focusing on the regulation of eosinophils by gut microbiota. During this time, he developed his current research program, which centers on understanding the role of diet and microbiota in mucosal immunity and beyond. In recognition of his scientific achievements as a postdoc, Dr. Arifuzzaman was awarded the 2024 Tri-Institutional Breakout Award for Junior Investigators. Dr. Arifuzzaman joined as tenure-track faculty member in the Division of Gastroenterology and Hepatology, Department of Medicine at Weill Cornell Medicine, where he is leading the cutting-edge research focused on defining the intricate tripartite relationships between nutrition, microbiota, and immunity in the contexts of infection, inflammatory and metabolic diseases, and cancer.
Distinctions:
- American Association of Immunologists ASPIRE Award
- Keystone Symposia Early-Career Investigators Travel Award
- Tri-institutional Breakout Prize for Junior Investigators
- K99 Pathway to Independence Award
- Kenneth Rainin Foundation Innovations Symposium Travel Award
- Intersections Science Fellows Symposium (ISFS) Associate
Selected publications:
1. Won TH*, Arifuzzaman M*, Parkhurst CN*, Miranda IC, Zhang B, Hu E, Kashyap S, Letourneau J, Jin WB, Fu Y, Guzior DV, JRI Live Cell Bank, Quinn RA, Guo CJ, David LA, Artis D, Schroeder FC. Host metabolism balances microbial regulation of bile acid signaling. Nature. 025 Jan 08. DOI: 10.1038/s41586-024-08379-9.
2. Arifuzzaman M✉, Won TH, Yano H, Uddin J, Emanuel E, Hu E, Zhang W, Li TT, Jin WB, Kashyap S, Grier A, JRI Live Cell Bank, Guo CJ, Schroeder FC, Artis D✉. Dietary fiber is a critical determinant of protective versus pathologic ILC2 responses and barrier inflammation. Journal of Experimental Medicine. 2024 May 06;221(5). PMID: 38506708.
- Featured on the cover of the issue.
3. Emanuel E*, Arifuzzaman M*, Artis D. Epithelial-neuronal-immune cell interactions: implications for immunity, inflammation, and tissue homeostasis at mucosal sites. Journal of Allergy and Clinical Immunology. 2024 May;153(5):1169-1180. PMID: 38369030.
- Featured on the cover of the issue.
4. Arifuzzaman M✉, Collins N, Guo CJ, and Artis D✉. Nutritional regulation of microbiota-derived metabolites: implications for immunity and inflammation. Immunity. 2024 Jan 09;57(1):14-27. PMID: 38198849.
- Featured on the cover of the issue.
5. Arifuzzaman M, Won TH, Li TT, Yano H, Digumarthi S, Heras AF, Zhang W, Parkhurst CN, Kashyap S, Jin WB, Putzel GG, Tsou AM, Chu C, Wei Q, Grier A, JRI IBD Live Cell Bank Consortium, Worgall S, Guo CJ, Schroeder FC, Artis D. Inulin fibre promotes microbiota-derived bile acids and type 2 inflammation. Nature. 2022 Nov 02;611(7936):578-584. PMID: 36323778.
- Highlighted in Nature Metabolism: Attwaters M. From inulin to inflammation. 2022 Nov 18; 4(11):1433.
- Highlighted in Cell Research: Cohen Y, Elinav E. Dietary fibers & immunity¾more than meets the eye. 2023 Jan 16.
*Co-first authors; ✉Co-corresponding authors
