
Research
I have a long-standing research program aimed at understanding the biological basis for sex differences in the brain. My research activities focus on two main systems: 1) cardiovascular circuitry in the hypothalamus; and 2) the learning and memory networks modulated by the hippocampus and its connections. These studies utilize multidisciplinary anatomical, molecular and physiological approaches including the technically sophisticated method of electron microscopic (EM) immunocytochemsitry.
Project 1. Neural mechanisms of hypertension susceptibility in a mouse model of menopause (in collaboration with Dr. Glass). We demonstrated that changes in NMDA-mediated excitatory signaling in hypothalamic paraventricular nucleus (PVN) estrogen receptor (ER) β -containing neurons are uniquely concomitant with slow-pressor angiotensin II (AngII) hypertension in females and males. Our EM studies showed that the redistribution of NMDA receptors in ERβ-containing neurons in response to slow pressor angiotensin II (AngII)-induced hypertension in females depends on age and hormonal state. Notably, the accelerated ovarian failure (AOF) mouse model revealed that hypertension susceptibility begins at peri-AOF which is a stage that mimics the erratic hormonal fluctuations seen in perimenopause. In peri-AOF females, AngII hypertension elevates the NMDA receptor GluN1 subunit on the plasmalemma of ERβ-containing neurons in a manner similar to that observed in males. We also found that changes in PVN NMDA plasticity in response to hypertension are greatly diminished at post-AOF which is a stage that mimics the negligible levels of estradiol seen in post-menopause. Importantly, our recent study shows that AngII-mediated hypertension increases plasmalemmal AMPA GluA1 in ERβ-containing PVN neurons only in post-AOF mice but not in peri-AOF females and males. These findings suggest that distinct neurobiological substrates underlie hypertension in peri- and post-menopausal periods.
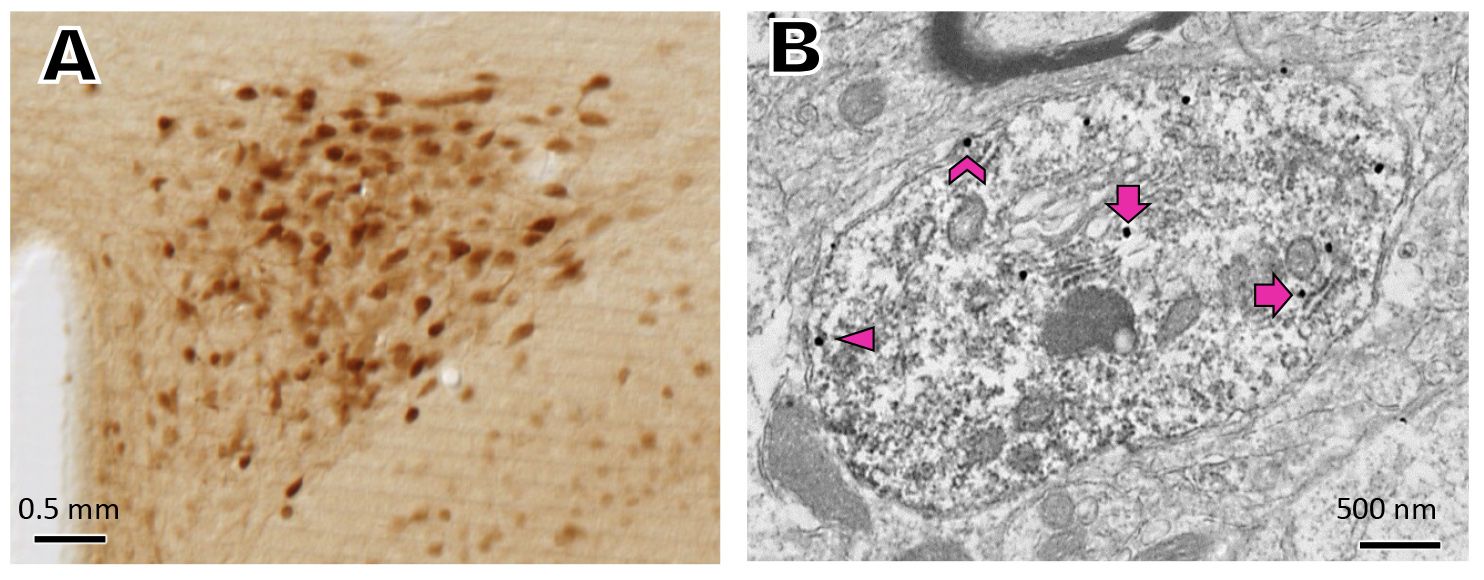
Figure 1 A. Estrogen receptor b (ERb)-EGP labeled neurons. B. Dendrite dually labeled for ERb (immunoperoxidase) and GluN1 (silver intensified immunogold; pink symbols). From Marques-Lopes et al. Neuroendo. 104: 239-256, 2017
Project 2. Sex differences in the hippocampal opioid system. The opioid circuit particularly the mossy fiber pathway, are crucial for associative memory processes important for addiction. Our anatomical studies, especially those utilizing EM immunocytochemistry and in situ hybridization, have provided unique insight into sex differences in the distribution of opioid peptides and receptors in specific hippocampal circuits and how these distributions are altered following stress and oxycodone-associative learning processes. We showed that opioid peptides and receptors are distributed within hippocampal circuits in females with elevated estrogen states in a manner that would enhance sensitivity to both endogenous and exogenous opioids. Moreover, we found that chronic stress primes the opioid system in females in a manner that would promote opioid-associative learning processes. In contrast, chronic stress has limited effects on the opioid system in males and thus reduces its capacity to support opioid-mediated learning processes. Interestingly, acute stress appears to prime males for opioid associative learning. On a broader scale the findings have important implications in understanding sex differences in opioid drug use and abuse. Currently, I have an grant (with Drs. Tilgner & Ndhlovu) characterizing the effects of chronic opioid exposure in single cell RNA expression in the non-human primate hippocampus following HIV.
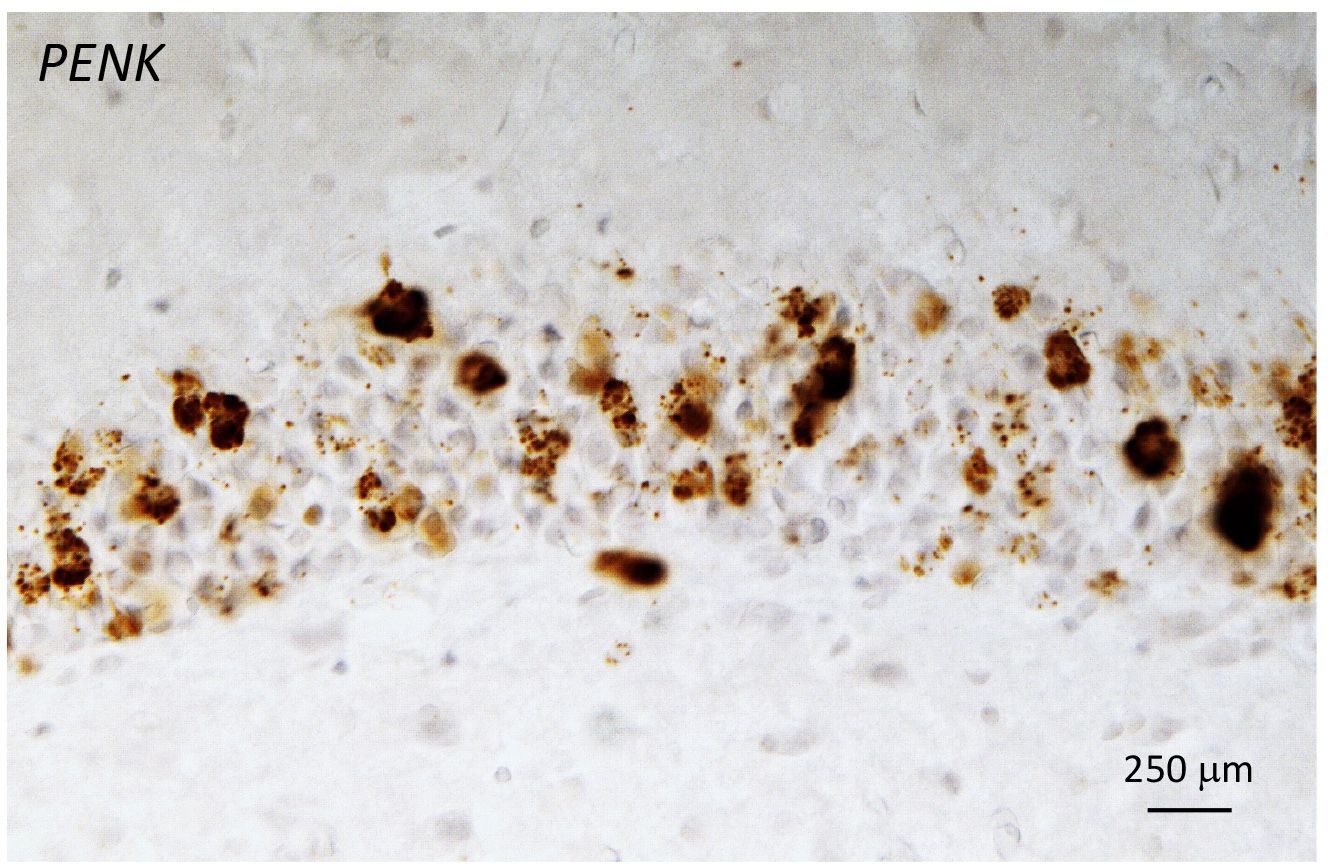
Figure 2 Hippocampus: PENK expression in the granule cell layer from a female rat following chronic stress. From Johnson et al JCN 529: 2636-2657 (2021)
Current Projects:
- Sex differences
- Hippocampus
- Hypothalamus
- Opioids
- Hypertension
- Neurodegeneration
Bio
Dr. Milner received her B.S. from the University of California, Irvine and her Ph.D. from the University of California, San Diego. She joined Weill Cornell Medicine in 1982 as a postdoctoral research fellow, and then rose through the faculty ranks to become a Professor of Neuroscience in 2003. She has been involved in graduate and medical school teaching as well as public outreach and diversity efforts for many years. Throughout her career, she has mentored more than 160 mentees, ranging from high school students to senior faculty, in the pursuit of science.
Distinctions:
- Excellence in Mentoring Award, Weill Cornell Medical College Postdoctoral Association (2008)
- Award for Teaching Excellence, Weill Cornell Medical College Medical School (for Brain and Mind) (2010)
- Excellence in Teaching and Mentoring Award, Weill Cornell Graduate School of Medical Sciences (2012)
- Dean’s Award for Excellence in Mentorship, Weill Cornell Medicine (2021)
- Forty Years of Service, Weill Cornell Medicine (2022)
