
Research
Our Mission
My lab investigates pathogenesis of age-dependent neurodegenerative diseases, like Alzheimer's disease and Parkinson's disease, at the cellular-molecular level. To study these diseases, we combine biochemistry, cell biology, mouse genetic models and mouse behavior.
Short-term Goals
Neurodegenerative diseases are associated with varied genetic and environmental factors, yet age remains a universal risk factor. Age-associated failure of protein homeostasis, or “proteostasis”, causes disease-linked proteins to misfold into non-physiological oligomers and aggregates, producing a myriad of cellular dysfunctions. Long lifetime of a neuron enhances the need for proteostatic mechanisms, such as molecular chaperones that stabilize native protein folds and degradation pathways that eliminate misfolded proteins. My lab is interested in the mechanisms that mediate proteostasis in neurons and how their failure over time leads to neurodegeneration.
We are investigating this topic by:
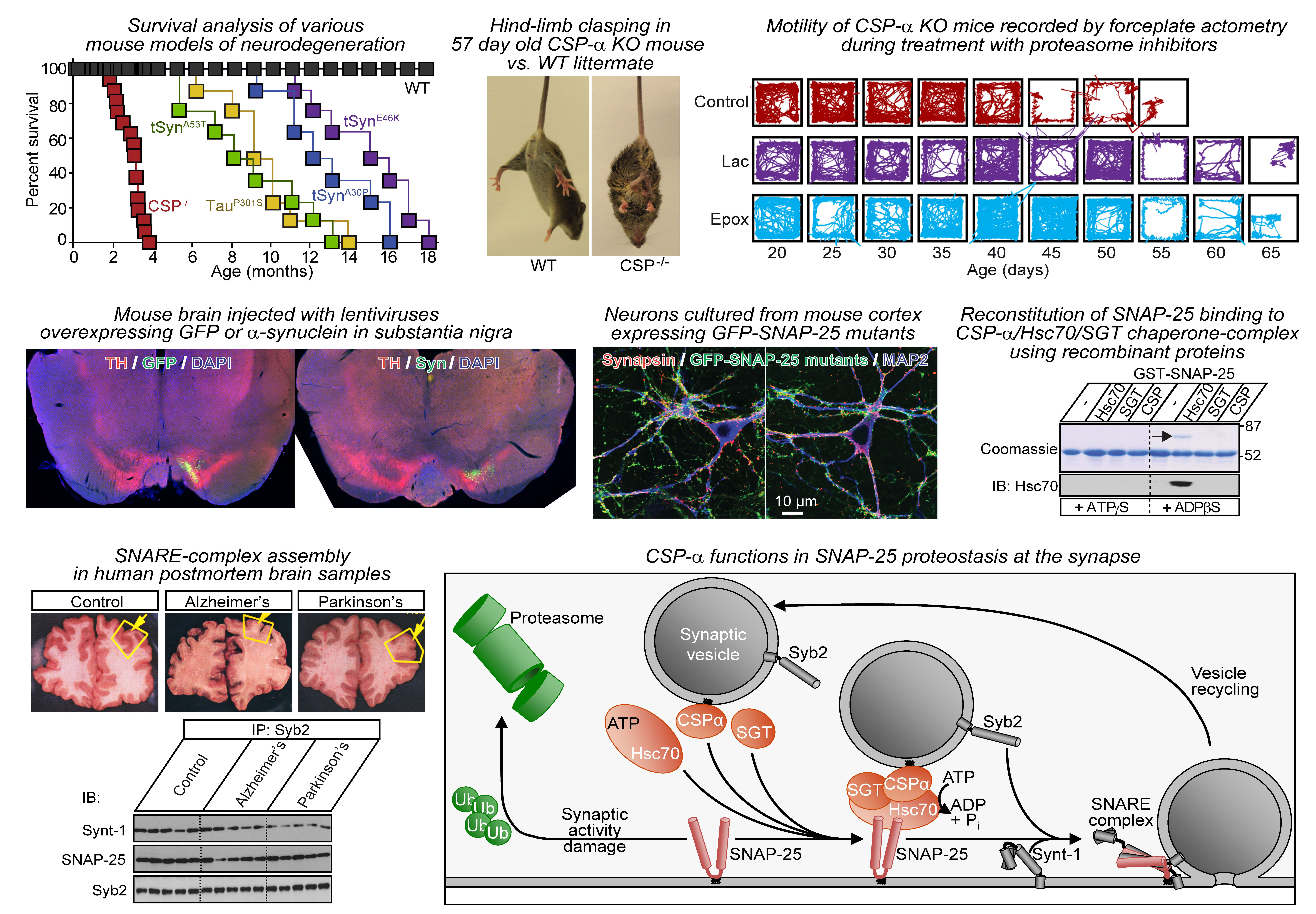
a) Studying how proteostasis of Tau protein is regulated by two co-chaperones of Hsc70: CHIP (C-terminus of Hsc/Hsp70 interacting protein) and CSP-α (cysteine string protein-α).
b) Investigating how neurodegeneration is caused by recently found human mutations in CSP-α which cause neuronal ceroid lipofuscinosis, an adult-onset neurodegenerative disorder.
c) Comparing lysosomes from brains of normal mice and from neurodegenerative mouse models to identify changes that accompany the decline of neuronal proteostasis.
Long-term Goals
Our research will uncover proteostatic mechanisms in neurons which allow them to live and function for a lifetime, and how these mechanisms fail in aging neurons. Understanding neuronal proteostasis in health and disease may lead to treatment strategies for multiple neurodegenerative disorders, like Parkinson’s disease and Alzheimer’s disease, which share age-dependent proteostatic defects.
Figure 1
Current Projects:
- Pathways that release aggregates of neurodegeneration-linked proteins from neurons.
- Modification of neurodegeneration by chaperone proteins, and their pharmacological manipulation.
- Pathobiology of tauopathies caused by traumatic brain injuries.
Bio
During Dr. Sharma’s PhD thesis with Dr. Gergely Lukacs at the University of Toronto and Hospital for Sick Children (Canada), he trained as a molecular cell biologist, while studying misfolding and quality-control of the cystic fibrosis protein CFTR. He applied this training to neuroscience during his postdoctoral training with Dr. Tom Südhof at UT Southwestern and Stanford University, where he acquired mouse genetics skills, to study animal models of human neurodegenerative diseases (Burré and Sharma et al., 2010 Science; Sharma et al., 2011 Nature Cell Biology). At Weill Cornell Medicine, the Sharma lab studies neurodegenerative disorders at the molecular-cellular level (Naseri et al. 2020 Nature Structural & Molecular Biology; Xie et al. 2022 Nature Communications).
Distinctions:
- Canadian Cystic Fibrosis Foundation Ph.D. Studentship
- Canadian Institutes of Health Research (CIHR) Doctoral Research Award
- Human Frontiers Long Term Fellowship, Human Frontiers Science Program (HFSP), Strasbourg Cedex (France)
- American Federation of Aging Research (AFAR) New Investigator in Alzheimer’s Research Award
- Alzheimer’s Association New Investigator Research Award.
- Sharma lab discovered the mechanism and use of Fe-chelators to treat Kufs Disease – a lysosomal storage disorder; use-patent pending (published in Naseri et al. Nature Structural & Molecular Biology, 2020; 27(2):192-201; first author was a PhD student in Sharma lab).
- Sharma lab generated a transgenic mouse line to specifically isolate neuronal lysosomes from mouse brains – Neu Lyso-Tag mice (published in Xie and Naseri et al. Nature Communications, 2022; 13(1):4918; both first authors were PhD students in Sharma lab).
- 2021 Mentor of the Year Award, from the Brain & Mind Research Institute and Neuroscience Graduate program, Weill Cornell Medicine.
