
Research
Projects include:
- Based on plasma concentrations, certain anesthetics can produce anesthetic, analgesic, and/or antidepressant actions. Anesthetics are potent modulators of brain activity and growth factors such as brain-derived neurotrophic factor (BDNF) mediate both local and global neural plasticity. However, BDNF signaling has divergent actions on neuronal survival, structure, and plasticity based on age, brain region, and activity. We are interested in exploring whether neurotrophic mechanisms converge on both antidepressant and anesthetic actions of ketamine and other mechanistically distinct anesthetics, and whether impaired BDNF signaling due to disease or genetic variation present additional vulnerability to deleterious neurocognitive effects of anesthesia.
Figure 1 Mouse hippocampal slice with viral infection of GCaMP6s in the granule cells of dentate gyrus.
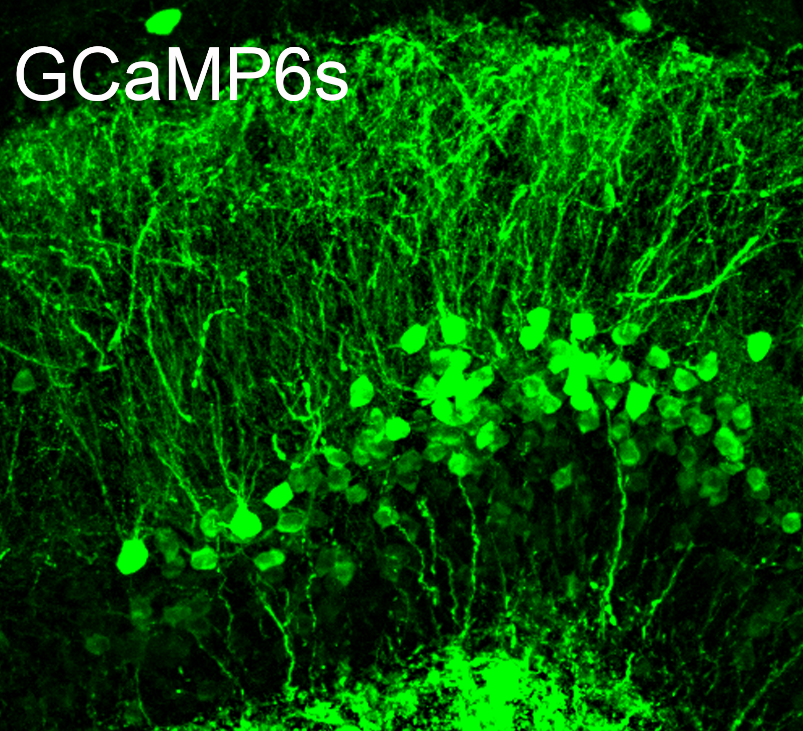
Figure 2 Neurostain
- Increased brain excitability is a prevalent feature of early Alzheimer’s Disease (AD), hastening cognitive decline and progression of disease. Variable expression of ion channels, such as sodium channels, influence neuronal excitability based on cell maturity. For example, different subtypes of sodium channels expressed in developing neurons are more susceptible to hyperexcitability compared to mature, established neurons. Interestingly, mature neurons, following injury or disease, can revert back to an earlier developmental window. We are interested in identifying whether neurons in early AD return to a ‘sensitive period’ of excitability and if volatile anesthetics, which potently inhibit sodium channels, can be utilized as novel therapeutics. The lab uses multiple transgenic mouse models to manipulate molecular and cellular brain circuitry. They combine several approaches including live hippocampal cell- and slice-imaging, fiber photometry, and in vivo calcium and optogenetic 2P imaging to understand clinically distinct, concentration-dependent effects of anesthetic agents.
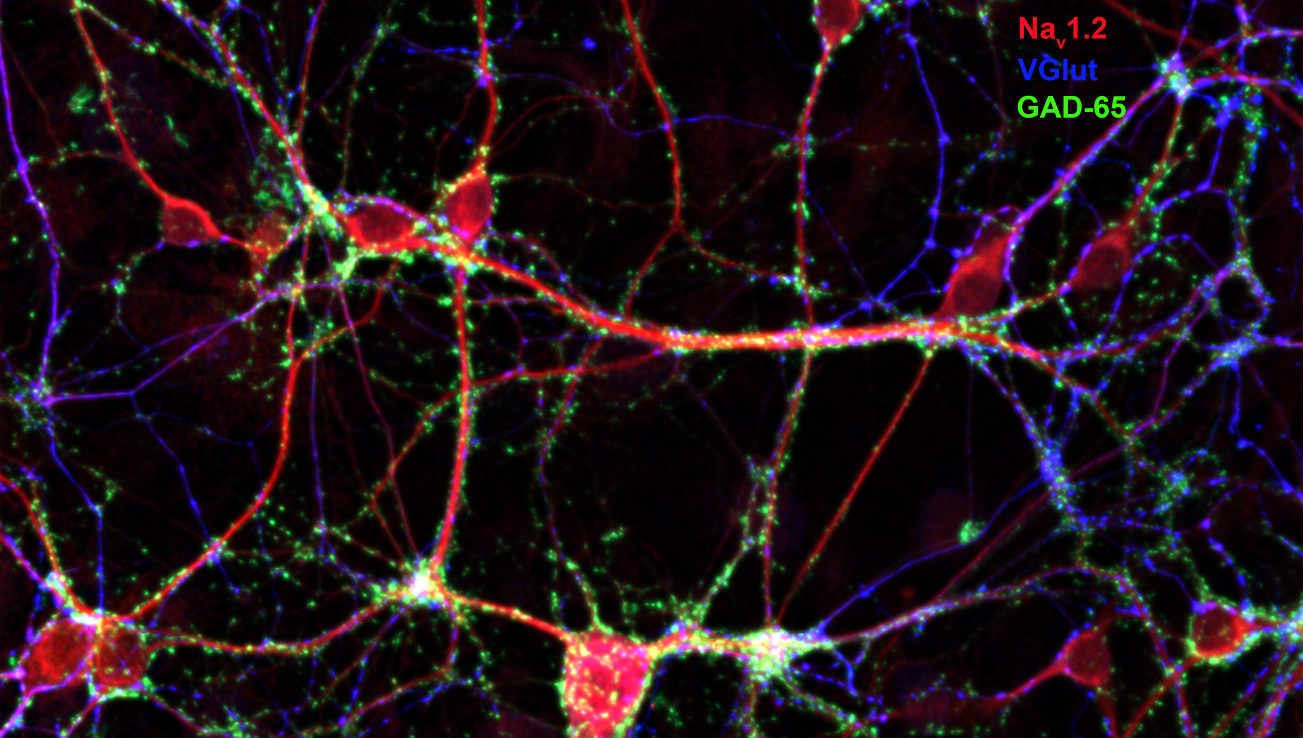
Figure 3 21 DIV hippocampal neurons transfected with egfp and immunolabelled for NAv1.2 (sodium channel); vGlut (marker for glutamate); and GAD-65 (marker for GABA).
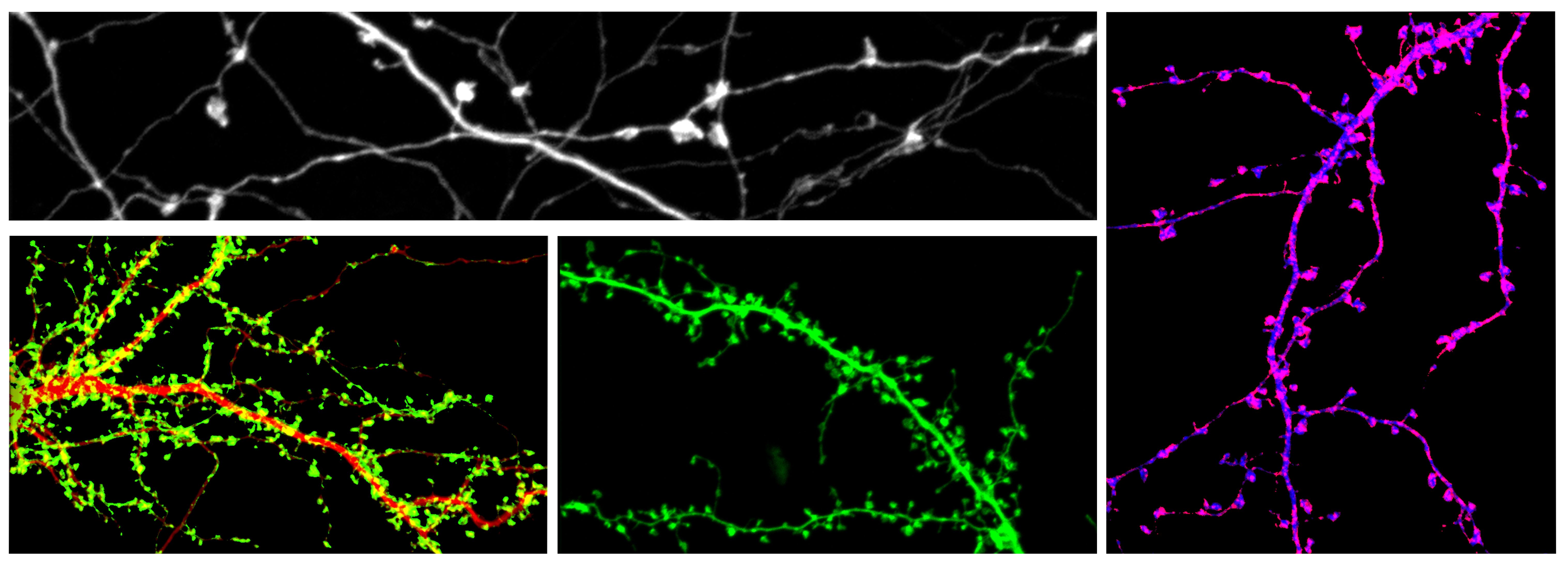
Current Projects:
- General anesthetics
- Neurotrophic signaling
- Depression
- Alzheimer’s Disease
- Synaptic Plasticity
Bio
Platholi received her Bachelors’ and Masters’ Degrees in Psychology from Stony Brook University and New York University, respectively, before attaining her PhD in Pharmacology at Weill Cornell Medicine. She conducted her postdoctoral research at University of
California, San Diego before returning to Weill Cornell Medicine as an Instructor and subsequently Assistant Professor in the Departments of Anesthesiology and Neuroscience.
