
Research
Adipose Biology in Cardiometabolic Health and Disease
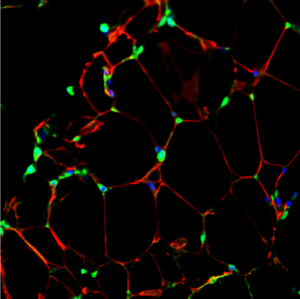
Aside from their well-known role in energy storage, adipocytes secrete many adipocytokines that can modulate systemic energy homeostasis and diseases such as diabetes and obesity. The Lo lab is interested in understanding how the adipose organ strikes this delicate balance between health and disease and harnessing the power of specific adipokines to prevent and treat diseases. They made the discovery that the adipokine adipsin communicates to pancreatic beta cells to prevent beta cell failure in type 2 diabetes. The Lo lab is also investigating how fat cells communicate with the heart to orchestrate systemic metabolic health.
Figure 1.
Pancreatic Islet Biology and Dysregulation in Diabetes
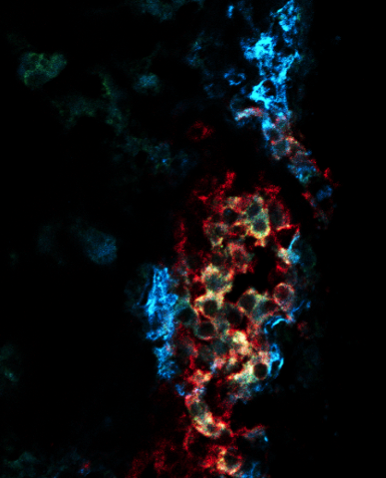
Insulin secreting beta cells are embedded within the pancreatic islets of Langerhans. Insulin is a key hormone that lowers blood glucose levels. In type 2 diabetes (where blood glucose levels are too high), there is dysfunction of the beta cells in insulin secretion. In advanced cases of type 2 diabetes there is more severe dysfunction and loss of beta cells to death and dedifferentiation. There are no current treatments that can arrest or reverse beta cell failure. We discovered that blocking the DUSP26 phosphatase may prevent beta cell death and dedifferentiation. There are also different subtypes of beta cells, including ones that are better insulin secretors. These high performing beta cells are lost in type 2 diabetes. Studies are underway to see how leveraging this knowledge can treat or prevent diabetes.
Figure 2.
Approaches
Single cell RNA-Seq, RNA-Seq, proteomics, phosphoproteomics, proximity labeling, metabolomics, spatial metabolomics, transgenic mice and animal models, pharmacological agents, samples from human specimens.
Current Projects:
- Adipose tissue function in health and disease
- Interorgan cross talk in cardiometabolic diseases
- Pancreatic beta cell failure and type 2 diabetes
- SARS-CoV-2 and metabolic dysfunction
- Metabolic stress of cardiovascular diseases
Bio
James Lo earned his Bachelor’s degree in biochemistry and M.D. and Ph.D. at the University of Chicago. In graduate school, he worked in the lab of Yang-Xin Fu and studied the lymphotoxin cytokine pathway in peripheral immune responses and control of lipid metabolism. He subsequently completed internal medicine residency at Massachusetts General Hospital and cardiovascular medicine fellowship at Brigham and Women’s Hospital and Harvard Medical School. For his postdoctoral work, he studied adipose tissue biology in the lab of Bruce Spiegelman at the Dana-Farber Cancer Institute and Harvard Medical School. In 2015, he started his lab at Weill Cornell Medicine.
Distinctions:
- Medical Scientist Training Fellowship
- Student Marshall, University of Chicago
- Gilead Sciences Research Scholar
- Feldstein Medical Foundation Award
- Irma T. Hirschl Career Scientist Award
- American Society for Clinical Investigation, Elected Member
