
Research
The Cesarman Lab focuses on studying the molecular pathogenesis of malignancies frequently associated with immunodeficiency, including AIDS, and more specifically the role of EBV and KSHV in the development of lymphomas and Kaposi sarcoma.
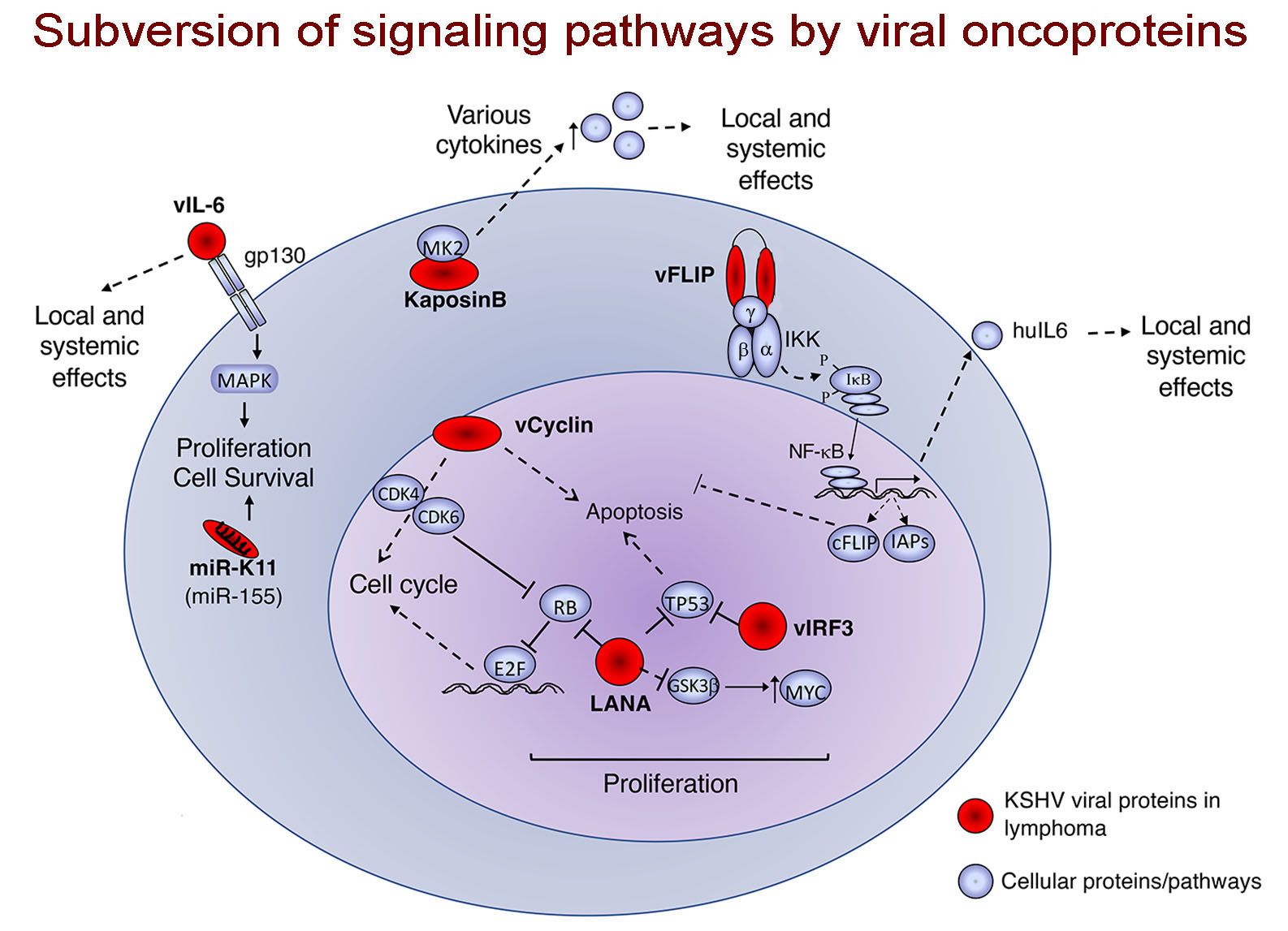
Cesarman’s research interest in lymphoma began as a graduate student working on the mechanism of MYC dysregulation in Burkitt lymphoma. This is a lymphoma type frequently associated with infection by an oncogenenic herpesvirus, Epstein-Barr virus (EBV). Notably, while most of us are infected by this virus, it also causes lymphomas, most frequently in people with immunodeficiencies (i.e. HIV infection, therapeutic immunosuppression, congenital immunodeficiency). While working on lymphomas associated with EBV infection and immunodeficiency, Cesarman contributed to the discovery of the causal agent of Kaposi sarcoma (KS), a virus called Kaposi Sarcoma Herpesvirus (KSHV; also known as HHV-8). KS is the most common cancer in people with HIV, and has been a major cause of AIDS-related mortality. KS is remains one of the most common cancers in Sub-Saharan Africa. Cesarman also identified the presence of KSHV as a defining feature of a lymphoma subtype, called primary effusion lymphoma. EBV and KSHV are the two known gamma-herpesviruses in humans, and both can infect B cells and lead to the development of B cell lymphomas, which are more frequent in the absence of a robust immune response. Interestingly, primary effusion lymphoma cells frequently contain the genomes of both KSHV and EBV, which may complement each other to cause this rare lymphoma type. Both of these viruses encode viral oncoproteins, that can subvert cellular signaling, and some of these are actually homologs to cellular proteins, probably pirated from the cellular genome throughout the course of evolution, maintaining some structural and functional similarities, but adapted to participate in the viral life cycle.
Figure 1
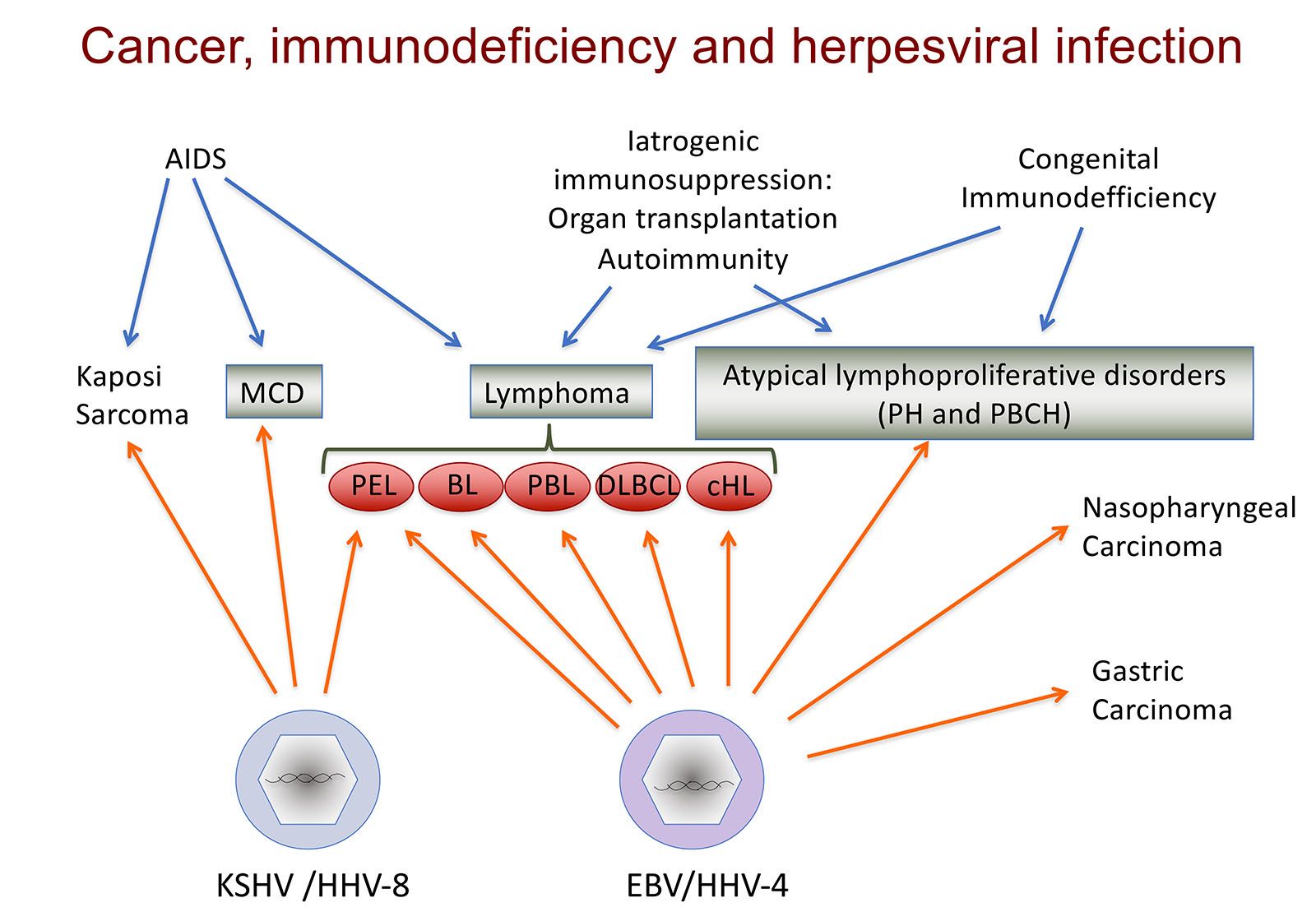
The Cesarman lab conducts work to investigate viral mechanisms of transformation and the virus-host cell interactions that are most relevant for carcinogenesis, which may provide opportunities for new treatment strategies. They have been been developing new diagnostic tools such as a point of care assay for Kaposi sarcoma. Work in the lab extends to malignancies that are only sometimes associated with viral infection, like diffuse large B cell lymphoma (DLBCL) and classical Hodgkin lymphoma (cHL). For example, her work includes deep sequencing of rare neoplastic cell populations, such as the Reed-Sternberg cells of cHL. These efforts led to the identification of recurrent mutations in linker histones (H1), and assessment of their functional significance. Work to identify new therapies for primary effusion lymphoma led the team to recognize a new role for adenosine kinase and purinergic signaling, which they have expanded to other high-grade lymphomas.
Figure 2
Current Projects:
- Modeling KSHV-associated cancers in vitro and in vivo
- Assessing molecular mechanisms of transformation in viral cancers
- Understanding the Kaposi sarcoma tumor microenvironment
- Identification of inhibitors of viral oncoproteins
- Adenosine signaling in lymphomas
- Role of linker histone mutations in lymphoma
- Combined role of EBV and HIV effects on the endogenous retrovirome
- Genomics of Kaposi sarcoma, viral lymphomas, and Hodgkin lymphoma
Bio
Cesarman was born in Mexico City, where she obtained her MD degree. She subsequently received a PhD from New York University, remaining in New York City since. She did a residency in Anatomic Pathology at the College of Physicians and Surgeons of Columbia University where she also completed her clinical training in Hematopatholgy. She has been faculty at Weill Cornell Medicine since 1994, working as a physician-scientist with a clinical practice in Molecular Pathology, while leading her research laboratory. Cesarman values her role as an educator and mentor to budding scientists and physicians at various career stages.
Distinctions:
- Discovery of a new cancer virus in Kaposi sarcoma (KSHV, also called HHV-8) (1995)
- Description of a new lymphoma type caused by KSHV (primary effusion lymphoma) (1995)
- Irma T Hirschl/Monique Weill- Caulier Career Scientist Award (2001)
- Society for Clinical Investigation (2001)
- Chair of the Weill Cornell General Faculty Council (2011-2013)
- Starr Cancer Consortium Awards (2007, 2007, 2015)
- Association of American Physicians (2015)
- David E. Hajjar Award for outstanding achievement in investigative pathology (2019)
