
Research
We live in an RNA world. Not only was RNA the original genetic code of life, recent years have witnessed remarkable impacts of RNA in the world at large. These include the Nobel prize for CRISPR/Cas9, the global human infection by a novel RNA virus, and subsequent worldwide transfection of humans with first-in-kind mRNA vaccines. Our lab takes integrative approaches to study diverse RNA programs, ranging from genetics and phenotypes, in vitro biochemistry, multiple genomic strategies, and recently, structural biology and single-molecule techniques. We take an evolutionarily broad perspective, including Drosophila and mammalian models, and non-model metazoans. Moreover, our studies are enabled by close collaborations with numerous colleagues, who continually take our work into new territories.
A major lab direction regards the mechanisms and biology of small (~20-30nt) regulatory RNAs, including miRNAs, siRNAs, and piRNAs (Figure 1). We revealed the conserved basis of target regulation by animal miRNAs, discovered a plethora of non-canonical small RNA biogenesis pathways, and elucidated the biological roles of small RNAs in development and behavior. These manifest in paradigms as varied as developmental patterning, stem cell progression, animal behavior, and meiotic drive. More broadly, we are excited that our work on fundamental small RNA mechanisms holds potentially broader impacts on experimental and therapeutic gene silencing, and we are now exploring such directions.
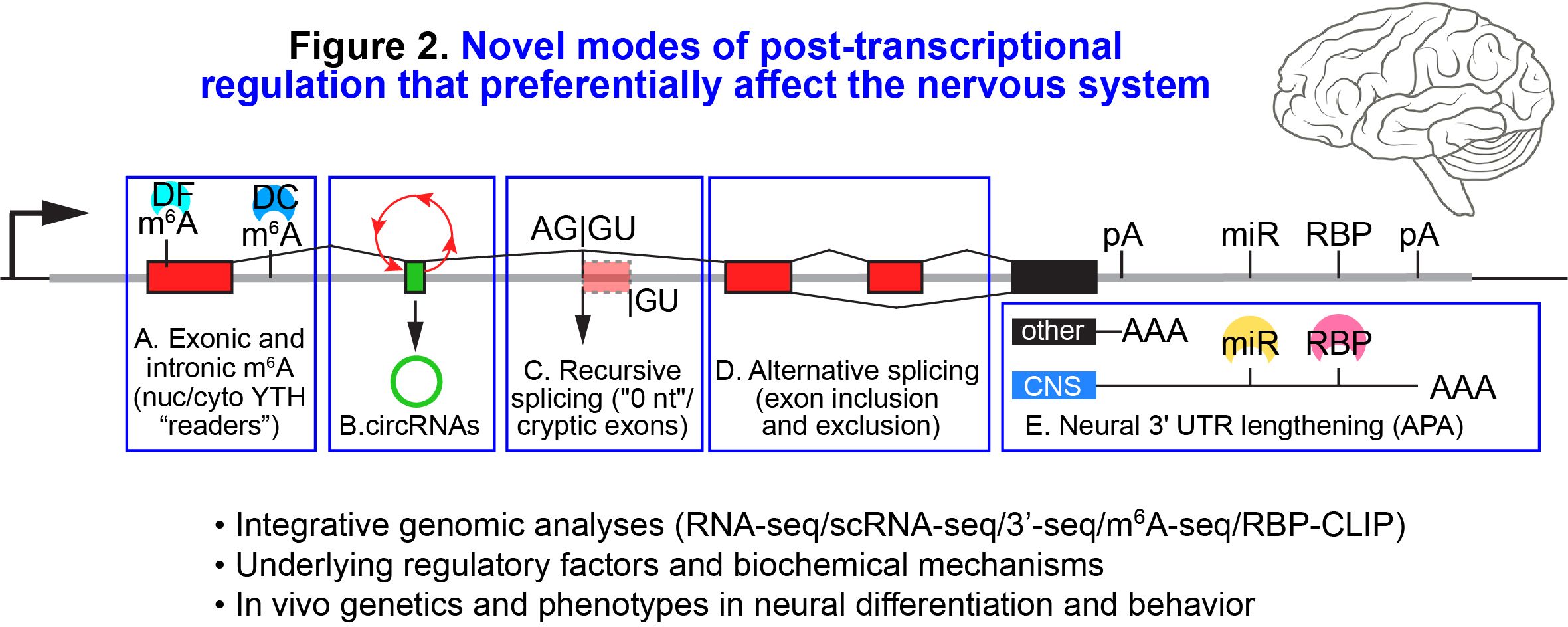
Another major effort concerns mRNA diversification by regulated splicing, alternative 3'UTRs, and RNA modifications (Figure 2). We use large-scale data integration, including single-cell RNA sequencing, to broadly extend the annotations of invertebrate and mammalian transcriptomes, including numerous non-canonical splicing isoforms, unexpected tissue-specific 3' UTR programs, and internal m6A modifications. We uncovered trans-acting factors that specific global neuronal-specific alternative splicing and 3' UTR programs, and showed their requirements in neural identity, germline stem cell dynamics, and adult behaviors. In the future, we hope to advance these efforts by incorporating CRISPR screening and directed differentiation of mammalian ESCs, as well as by exploring disease and cancer settings.
Finally, we maintain long-standing interests in cell signaling via the Notch pathway, and gene regulation by transcription factors. These efforts include the discovery of the BEN domain as a structurally novel DNA binding domain that defines a previously unrecognized family of transcription factors in invertebrates and mammals. Our most recent studies utilize crystal structures and Alphafold to extend this family, and current efforts will dissect BEN factors in controlling hESC gene expression and heterochromatin.
Figure 1
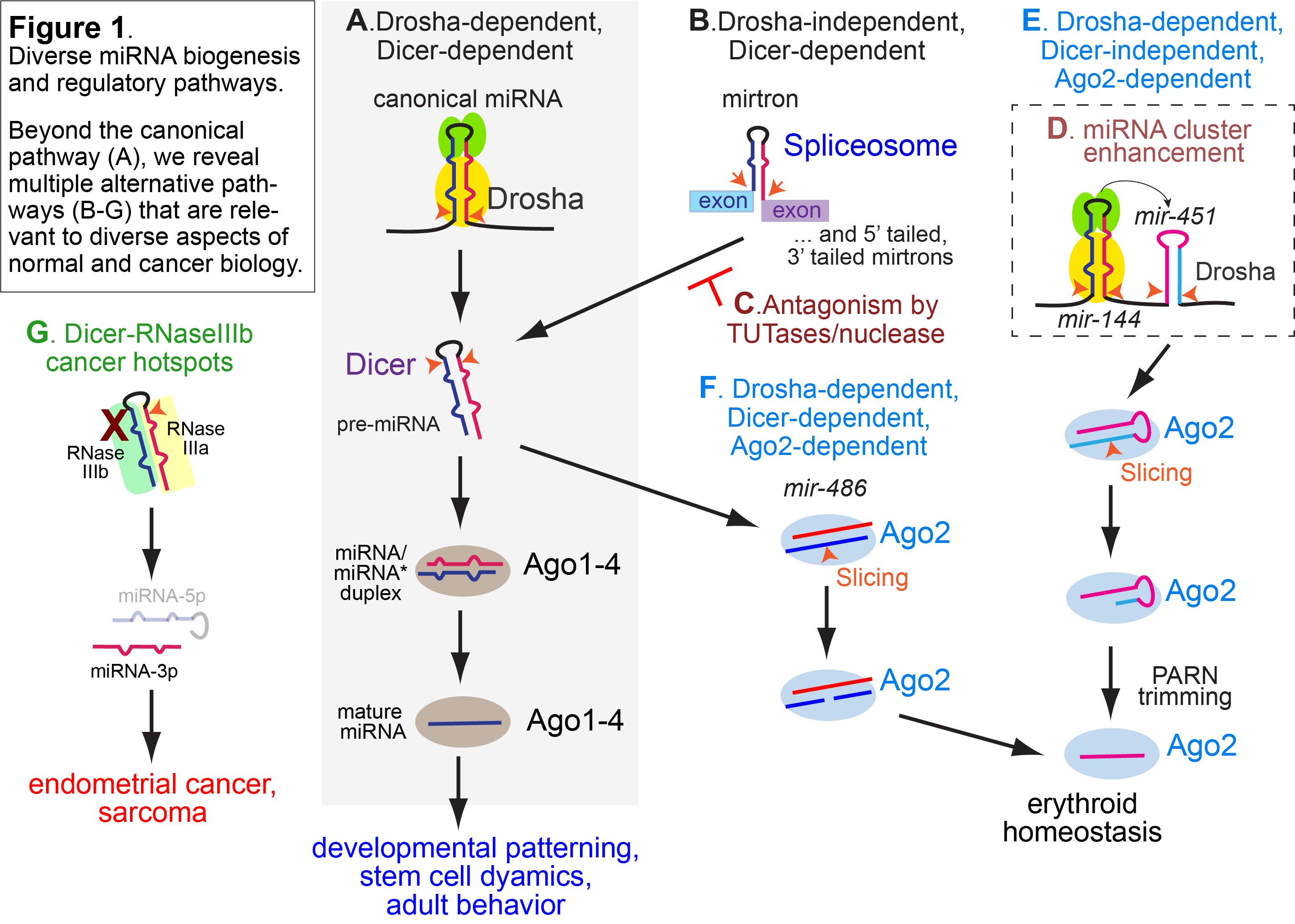
Figure 2
Current Projects:
- Biogenesis and biology of miRNAs in Drosophila and mammals
- Therapeutic applications of regulated small RNA production
- Generating the neural-specific transcriptome during development and behavior
- Intragenomic conflict, endogenous RNAi and speciation
Bio
Dr. Eric Lai obtained his B.A. from Harvard, where thesis research with Dr. Gary Ruvkun exposed him to developmental patterning and post-transcriptional regulation by small RNAs. He continued these themes during his Ph.D. with Dr. James Posakony (UC San Diego) and postdoc with Dr. Gerald Rubin (UC Berkeley), during which he elucidated novel components of Notch signaling, and uncovered the regulatory logic of miRNAs. Since joining the Developmental Biology Program at Sloan-Kettering Institute in 2005, Dr. Lai has pursued novel mechanisms of transcriptional and post-transcriptional regulation and their biological requirements, focusing on the nervous system and germline of Drosophila and mammals.
Distinctions:
- 2009 Louise and Allston Boyer Young Investigator Award for Basic Research
- 2008 New York Academy of Sciences-Blavatnik Award for Young Scientists, Finalist
- 2007-2009 Kimmel Scholar of the Sidney Kimmel Foundation for Cancer Research
- 2006-2008 V Scholar of the V Foundation for Cancer Research
- 2005 Career Award in the Biomedical Sciences, Burroughs Wellcome Fund
- 2004-2006 Special Fellow of the Leukemia and Lymphoma Society
- 2000-2003 Fellow of the Damon Runyon Cancer Research Foundation
- 1993 Phi Beta Kappa, Harvard College
