Using cutting-edge techniques, Weill Cornell Medicine and Memorial Sloan Kettering investigators have visualized the structure of a receptor targeted by an anti-cancer immunotherapy. The new information may help scientists improve this type of cancer treatment.
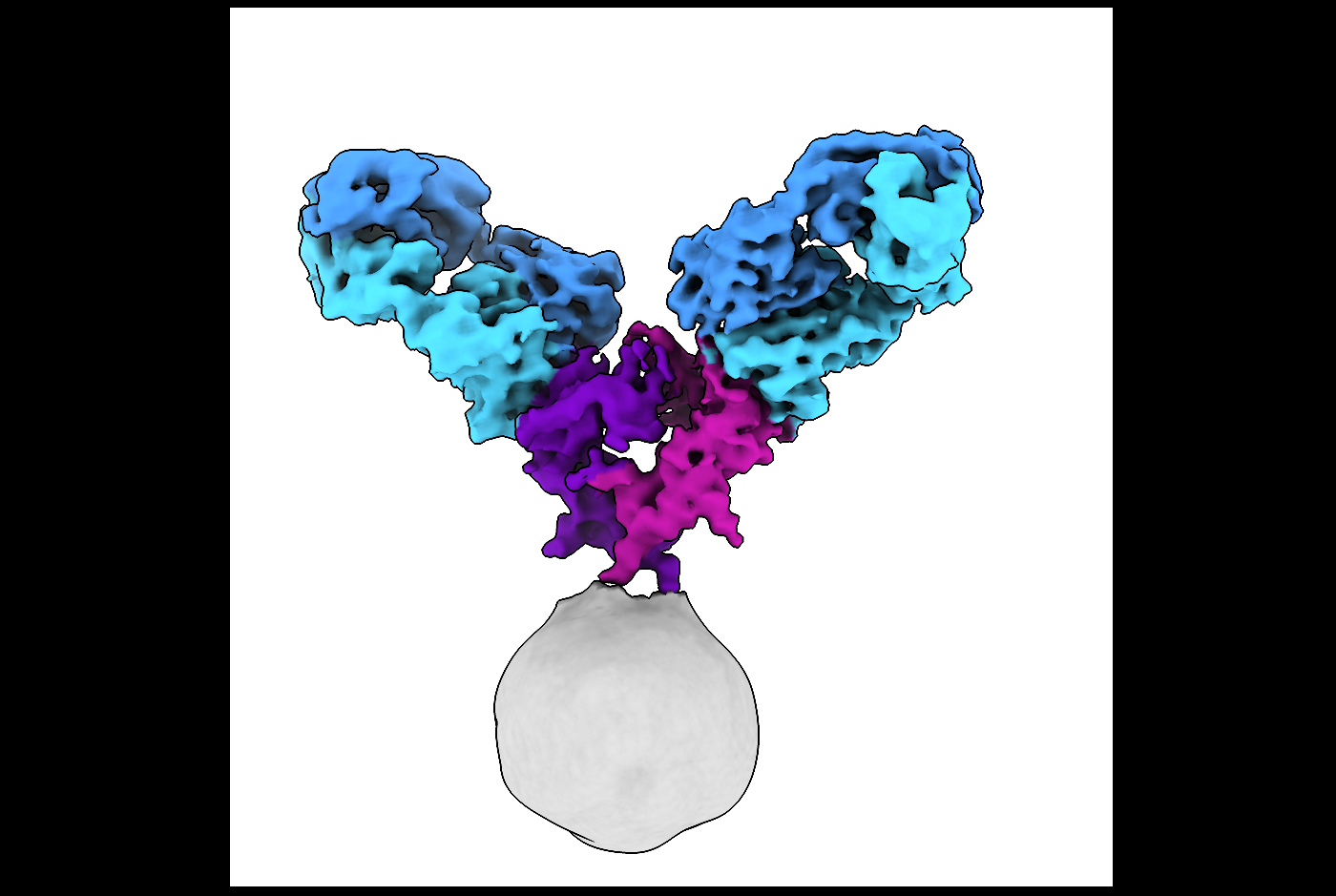
The study, published Feb. 25 in Science Advances, used cryo-electron microscopy (cryo-EM) to reveal the full-length structure of the glucocorticoid-induced tumor necrosis factor receptor (GITR), an advance over previous studies that examined fragments of the protein. The work also shows how the receptor interacts with a therapeutic antibody and the receptor’s naturally occurring protein partner called a ligand. The approach used may facilitate similar studies of other tumor necrosis factor (TNF) receptors, which play a crucial role in the immune system and human health.
“The TNF receptor family of receptors is large and their structures have been studied for around 30 years using X-ray crystallography,” said co-senior author Dr. Joel Meyerson, assistant professor of physiology and biophysics at Weill Cornell Medicine. “This past work provided valuable information about portions of the receptors. Using cryo-EM, we were able for the first time to look at a full-length TNF receptor.”
When GITR is activated, it turns off immune cells that suppress the immune response. Thus, GITR is a promising target for cancer immunotherapies that aim to activate the immune responses that destroy tumors. Preclinical studies of antibodies targeting GITR have shown they can successfully treat cancers of the lymph nodes, skin and colon. Now, clinicians are testing anti-GITR therapies in human clinical trials.
“Tumor necrosis factor receptors are important targets for anti-tumor cancer therapies,” explained the study’s co-lead author Changhao He, a doctoral candidate in the Weill Cornell Graduate School of Medical Sciences. “Studying how they are naturally activated or how anti-TNF antibody treatments activate them is crucial.”
Using cryo-EM, the team revealed that the intact GITR receptor forms from two copies of the crescent-shaped GITR protein. Solving the structure of the receptor with an antibody yielded new insights. The team also compared the activity of antibody and ligand.
“We were able to show that the antibody actually mimics the ligand to drive immune signaling,” Dr. Meyerson said. Both the ligand and the antibody cause the receptors to cluster together.
“Immunologists believe that clustering of receptors enhances downstream signaling,” he said. “Our study provides evidence supporting this hypothesis.”
“Understanding the mechanism behind the activity of drugs in clinical development is key to ensuring their therapeutic success,” said co-senior author Dr. Taha Merghoub, professor of immunology research in medicine at Weill Cornell Medicine and a cancer immunologist in the Ludwig Collaborative Laboratory at Memorial Sloan Kettering. “Working collaboratively across multiple disciplines such as we have done with Dr. Meyerson will ensure faster development of mechanism-based approaches to improve current and potential future therapeutic interventions.”
Now that Dr. Meyerson and his colleagues know that clustering of the receptors can drive immune signaling, they want to learn more about how it works. They also plan to use cryo-EM to study more TNF receptors to determine if they also use this signaling mechanism or different ones.
It also may offer new insights that drug developers can use to enhance or test these therapies. “Structural biology is becoming more and more a part of therapy development,” Dr. Meyerson said. “I hope this study gives drug developers another angle to consider.”
